Cecal Carcinoma following autologous transplantation for Multiple Myeloma.
Καρκίνος Τυφλού μετά ΑΜΜΟ για Πολλαπλούν Μυέλωμα.
Introduction
Multiple Myeloma is a malignancy of plasma cells characterized by the rapid proliferation of plasma cell clones, resulting in the production of monoclonal immunoglobulins. Although it is typically considered an incurable disease, advances in therapeutic options have led to significant improvements in patient survival. Novel therapies include autologous transplantation and innovative agents such as immunomodulatory drugs and proteasome inhibitors (cellular structures responsible for protein degradation).
The administration of high-dose Melphalan as monotherapy was the most common treatment regimen (81%). From 1990 to 2010, newer agents were used prior to transplantation in 69% of patients, including Thalidomide in 34%, Lenalidomide in 14%, and Bortezomib in 21%. Following transplantation, maintenance therapy included thalidomide (15%), lenalidomide (11%), bortezomib (9%), and Interferon (6%). Most patients (59%) underwent transplantation 6–12 months after diagnosis, 27% within 6 months, and 14% between 12 and 18 months after diagnosis.
Immunomodulatory agents are now considered an integral part of the comprehensive treatment of multiple myeloma. As increasing evidence accumulates regarding their efficacy, more is also being learned about the risks associated with their use. In particular, several reports support an increased risk for the development of secondary primary malignancies (SPMs) among patients treated with these agents. Given the continuous improvements in survival resulting from these therapeutic regimens, patients with multiple myeloma are expected to survive longer, thereby allowing more time for the development of such malignancies.
Since multiple myeloma is not considered a curable disease—with the rare exception of allogeneic stem cell transplantation—healthcare providers are increasingly faced with the dilemma of selecting the most appropriate treatment for patients with multiple myeloma who subsequently develop secondary primary malignancies, as therapeutic options often demonstrate very limited overlapping efficacy.
Therefore, it is important to determine whether the increased risk of developing new cancers is attributable to the disease itself or to its treatment. Several, though not all, studies support an increased risk of developing new malignancies, including Acute Myeloid Leukemia (AML), Myelodysplastic Syndrome (MDS), and secondary primary malignancies (SPMs) in patients with multiple myeloma, regardless of whether they underwent autologous transplantation.
A new cancer is defined as a previously undetected invasive malignancy that develops following transplantation for the treatment of multiple myeloma. In situ cancers and other precancerous abnormalities (e.g., squamous intraepithelial neoplasia) are excluded.
We present the case of a patient who developed invasive adenocarcinoma of the cecum following autologous transplantation while receiving induction therapy.
Case Report
A 67-year-old male patient who was diagnosed with multiple myeloma in 2017 was initially treated with the administration of lenalidomide, bortezomib, dexamethasone, and zoledronic acid. A partial response was then achieved according to the criteria of the International Myeloma Working Group (IMWG).
The patient was considered a good candidate for autologous transplantation, as he was in excellent general condition, exhibited no organ dysfunction, and, finally, successful stem cell collection was achieved. After 6 cycles of the initial therapy, he was switched to the daratumumab, pomalidomide, and dexamethasone regimen, with the hope of achieving the best possible response prior to autologous stem cell transplantation. The transplantation was performed successfully, and the patient was under close hematological follow-up.
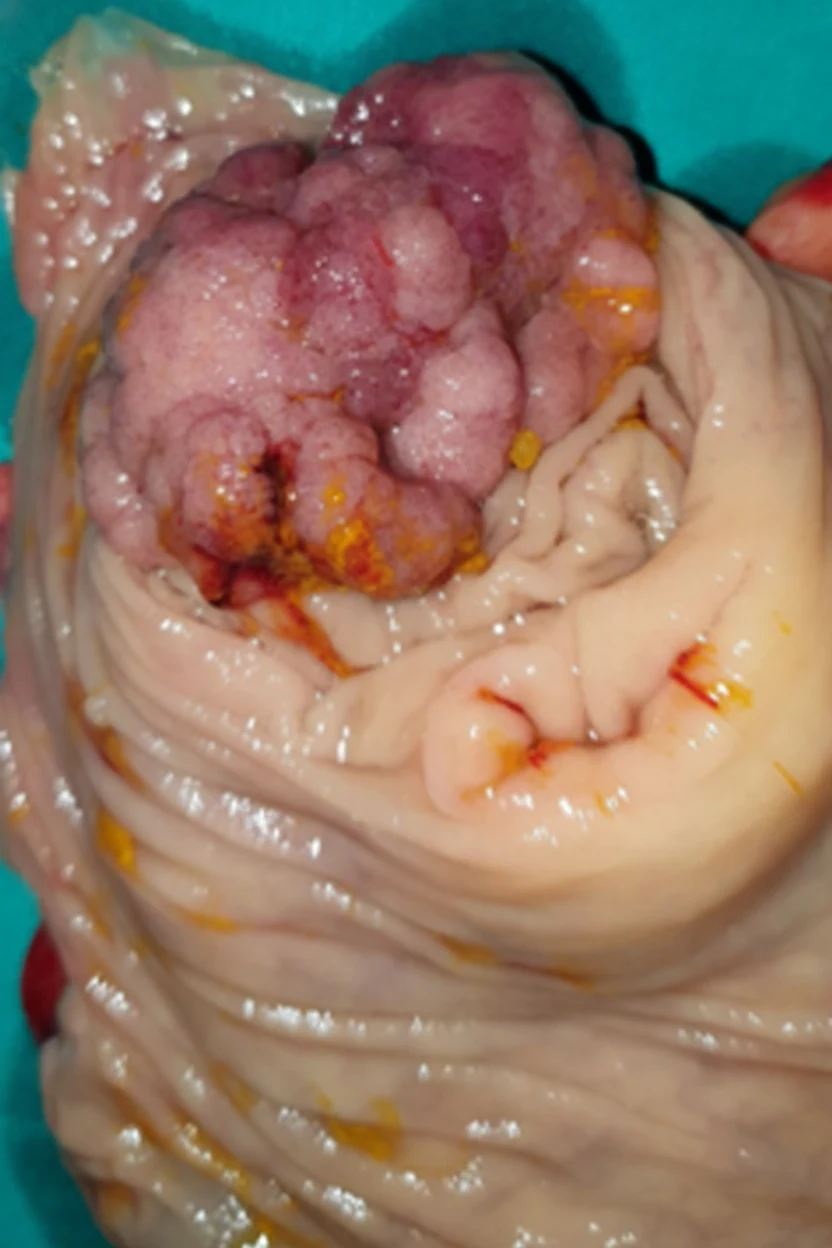
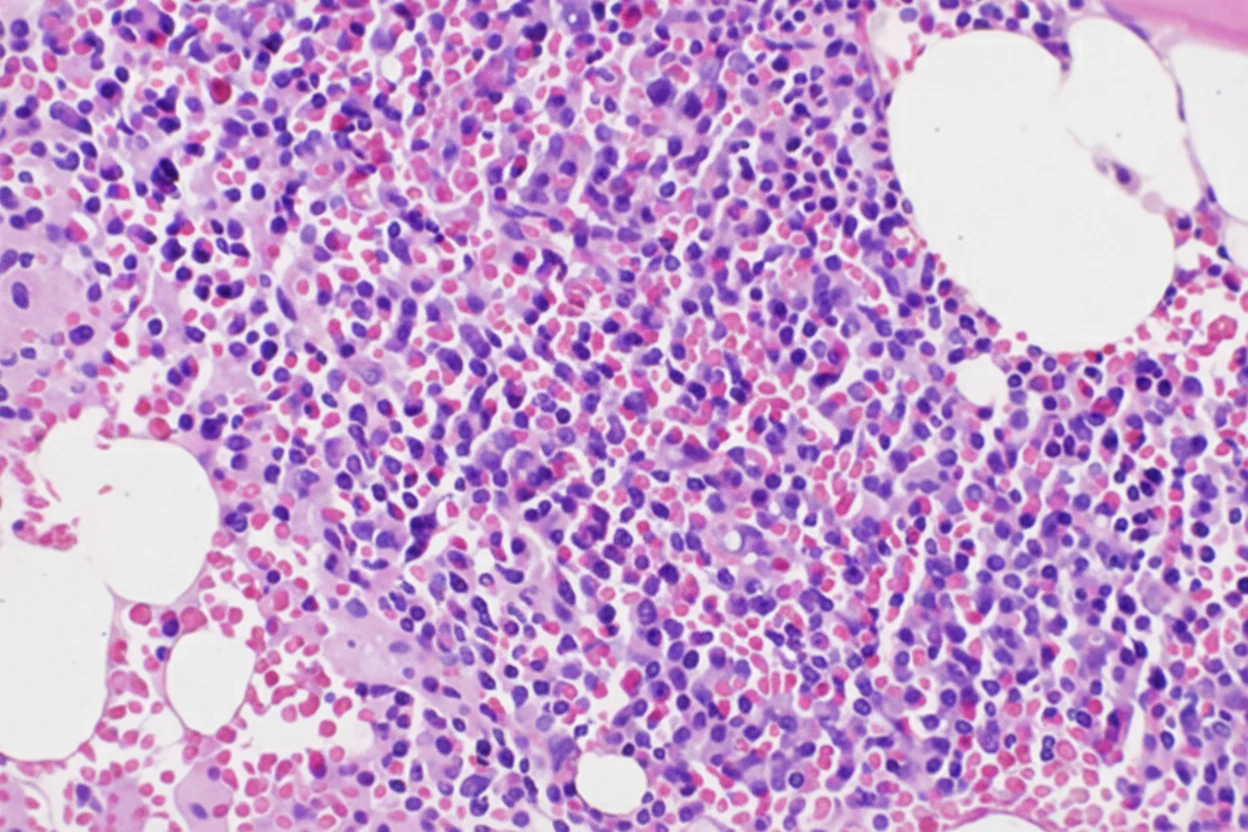
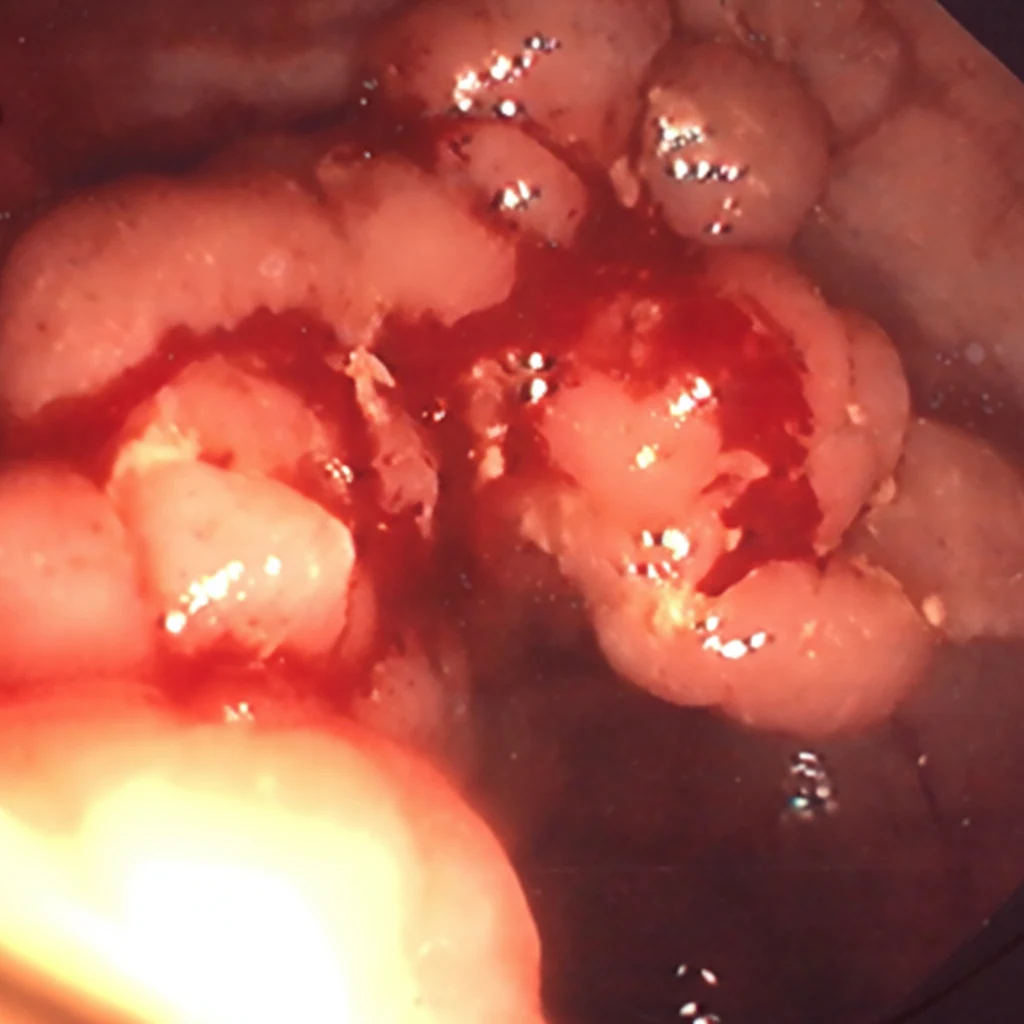
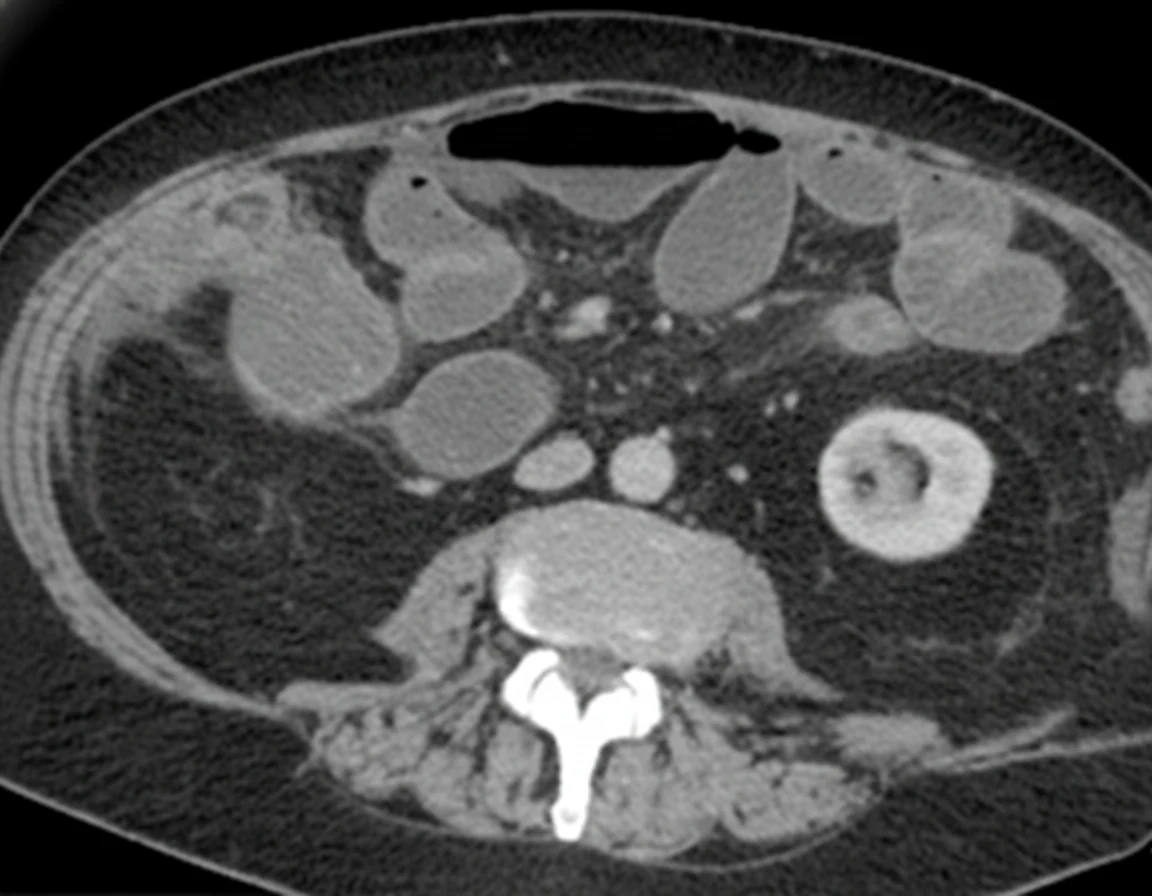
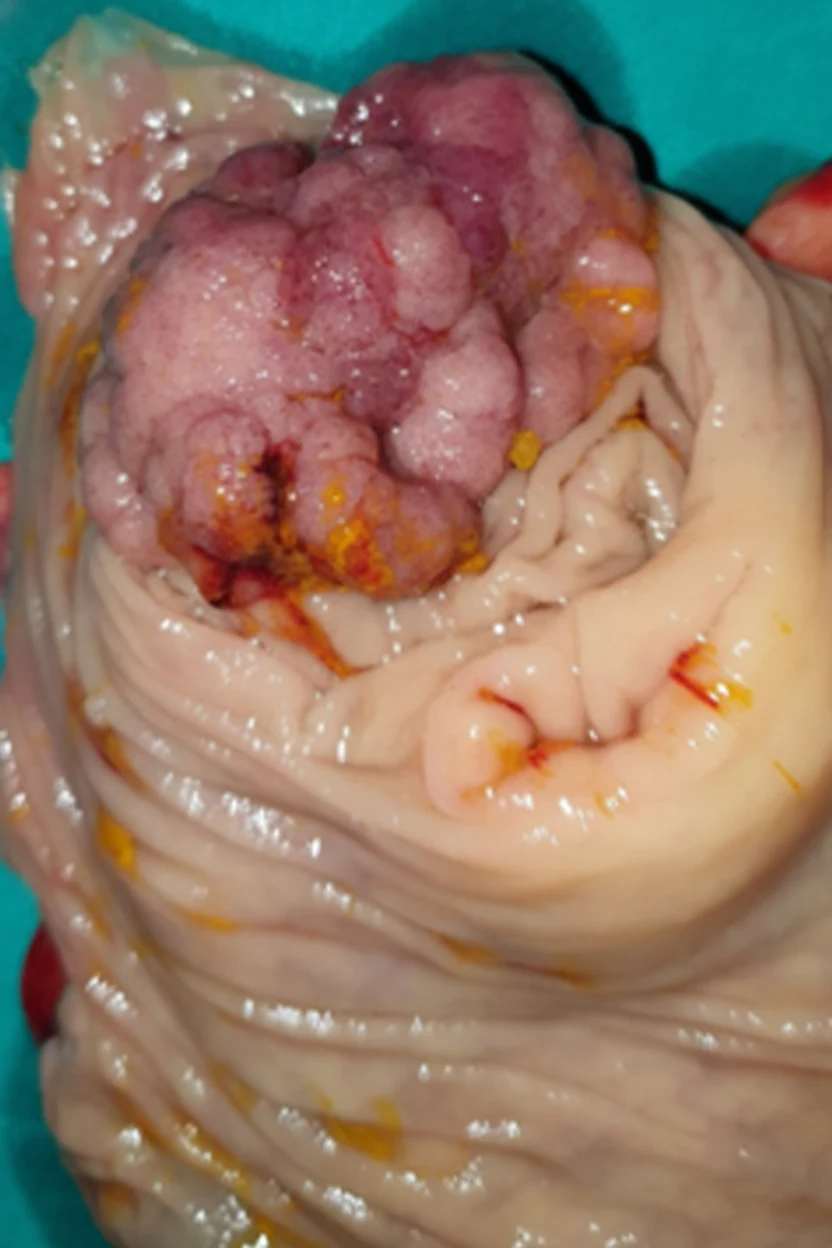
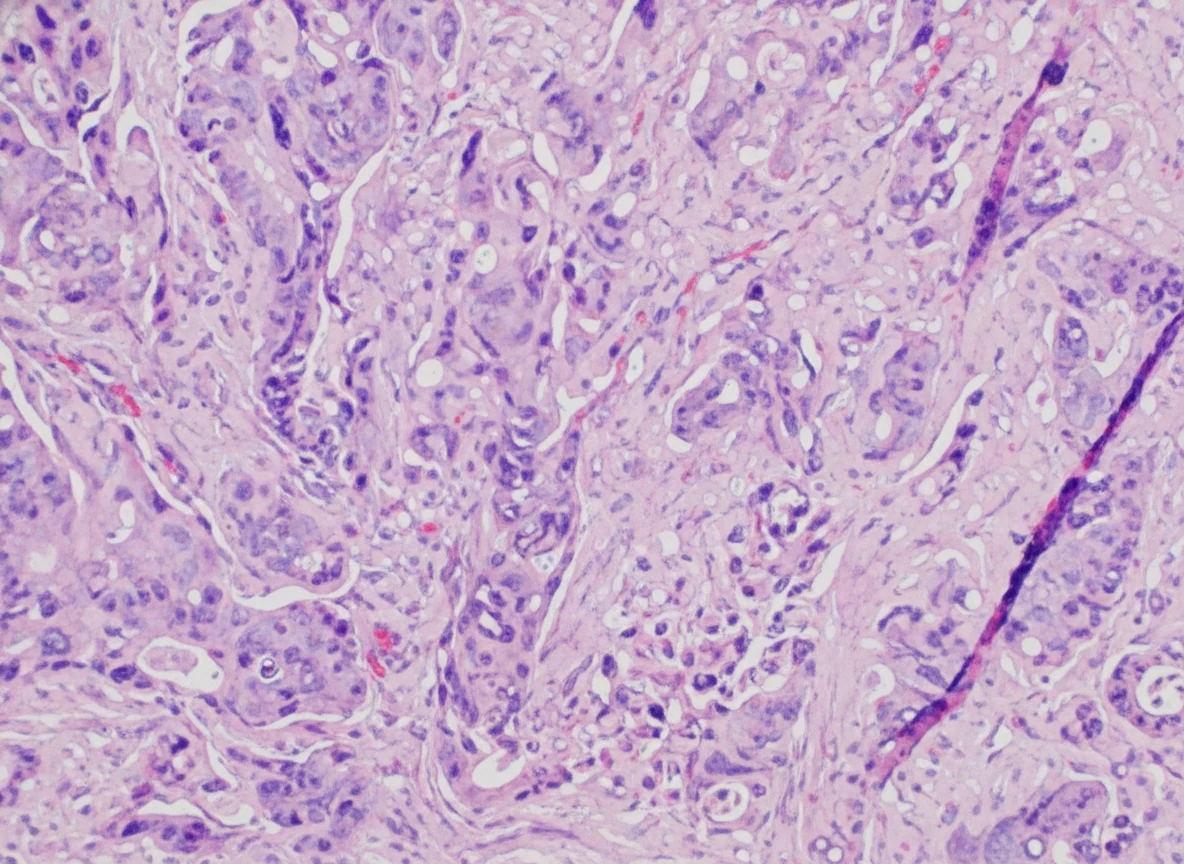
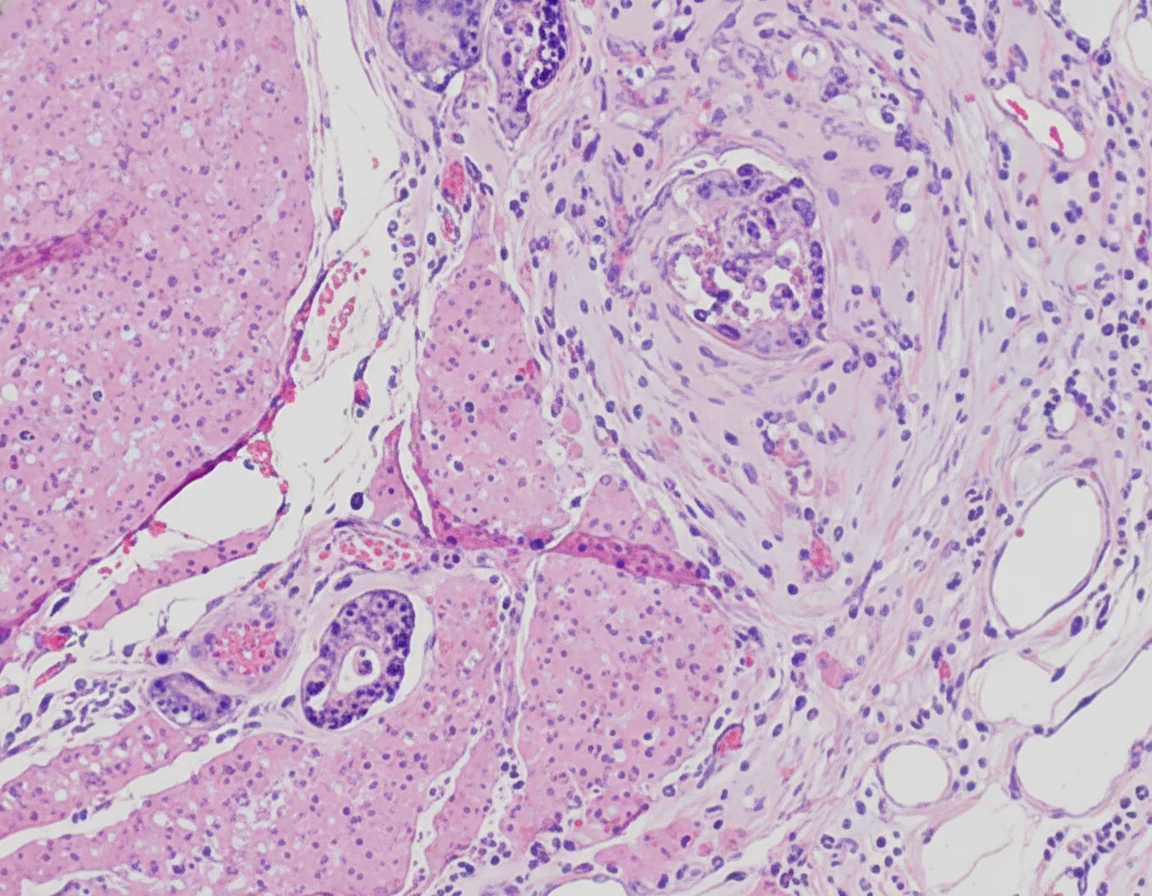
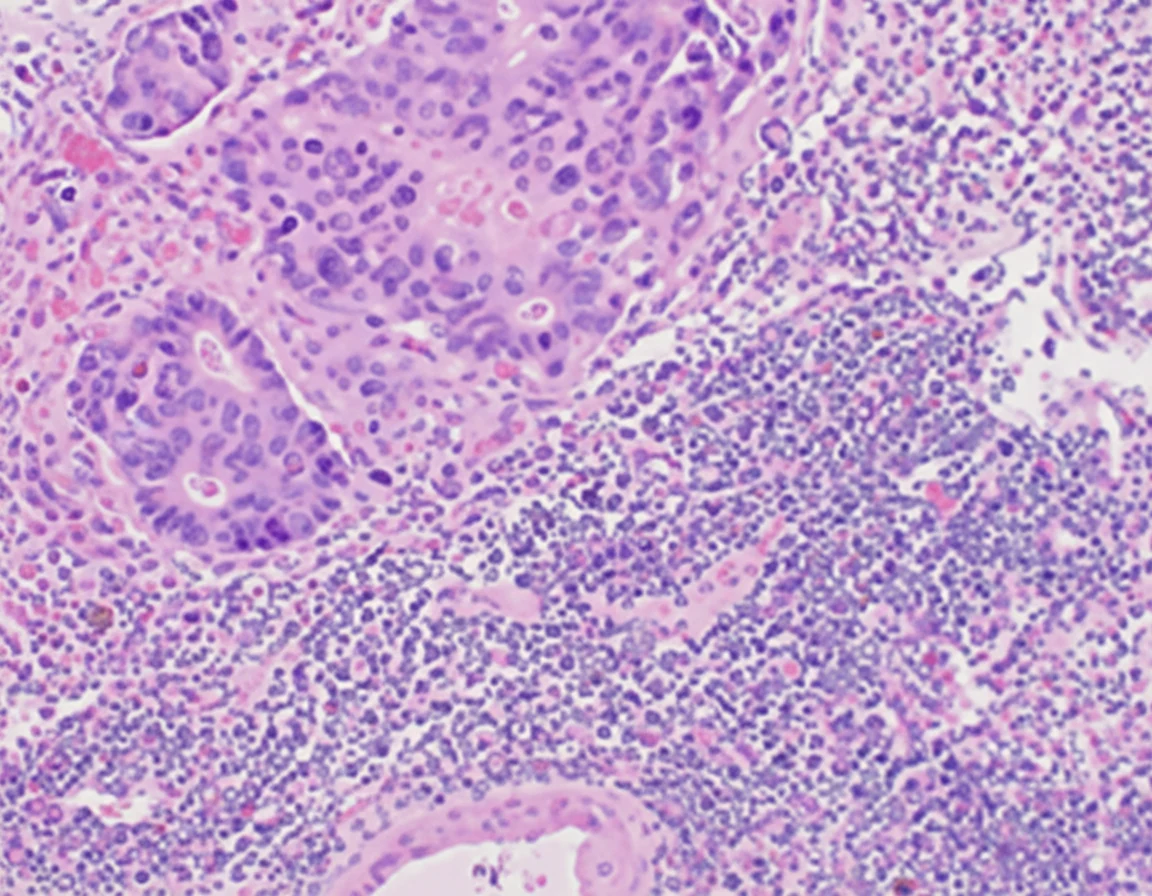
In 2019, while the initial disease was in remission, he complained of abdominal pain and passage of dark-colored stools. Abdominal computed tomography revealed the presence of a probably neoplastic cecal lesion.We then performed a colonoscopy, which revealed the presence of a mass approximately 6.5 cm in size, from which multiple biopsies were obtained, establishing the diagnosis of a moderately differentiated adenocarcinoma.
Of note, the patient had undergone a colonoscopy in 2016, during which two benign hyperplastic polyps had been removed from the descending colon.With the concurrence of the hematologists, we performed a right hemicolectomy.Disease staging was IIIB (pT3N2aM0).
The patient had an uneventful postoperative course and was discharged from the hospital 8 days later. With the concurrence of oncologists and hematologists, he began adjuvant therapy consisting of capecitabine and continuation of bisphosphonate therapy for 6 months.
During this period, the myeloma status remained stable, with monoclonal M-spike protein levels of 0.25 g/dL. He subsequently resumed myeloma-directed therapy with daratumumab, pomalidomide, and dexamethasone.
Discussion
Advances in the therapeutic management of multiple myeloma, including the combination of high-dose melphalan and autologous stem cell transplantation, as well as the introduction of novel immunomodulatory antimyeloma agents and proteasome inhibitors, have increased the life expectancy of patients with multiple myeloma.
However, with prolonged survival, the management of clinical conditions associated with increased longevity, such as secondary primary malignancies (SPMs), has become clinically relevant.
Secondary primary malignancies primarily manifest as hematological malignancies, with a 7-fold increase in the incidence of acute myeloid leukemia following the diagnosis of multiple myeloma.However, solid secondary primary malignancies have been studied and reported in a series of 36,491 cases of multiple myeloma from the Surveillance Epidemiology and End Result (SEER) Program between 1973 and 2008, with a heterogeneous distribution of risk based on the specific solid subtype.
The relationship between the treatment of multiple myeloma and secondary primary malignancies was first identified in 1996 by Govindarajan et al., after the administration of high-dose chemotherapy, particularly melphalan. Additionally, Krishnan et al. reported an 11.2% increased risk of developing secondary primary malignancies 10 years after autologous stem cell transplantation.With the evolution of immunomodulatory drugs, an association was found between lenalidomide and secondary primary tumors, particularly in patients who received oral melphalan.
The development of secondary primary malignancies is likely multifactorial, encompassing the treatment of multiple myeloma, the tumor microenvironment, and genetic and environmental factors.
The mechanism of action of lenalidomide is complex, and its ability to promote the development of SPM remains unclear. The immunosuppressive effects of the drug on the tumor microenvironment may lead to the escape and/or growth of abnormal oncogenic clones, resulting in the development of an SPM.Facilitation of SPM development may arise, as lenalidomide impairs nucleotide excision repair mechanisms following DNA damage induced by melphalan.
The risk of developing colorectal cancer is increased by 50% at 5 years after the diagnosis of multiple myeloma and may be attributable to shared immunological abnormalities of the two diseases.It is difficult to ascertain whether any specific factor contributed to the development of cecal carcinoma in our patient after receiving treatment and transplantation for multiple myeloma, or whether the emergence of the carcinoma could simply be attributed to his age.At this point, we would like to recall that in 2016, two hyperplastic polyps had been removed colonoscopically from the patient, while in 2014 the screening colonoscopy performed had been entirely normal.
Additionally, it must be emphasized that during adjuvant therapy with capecitabine for cecal carcinoma, therapy for the management of multiple myeloma was suspended, except for zoledronic acid, and, despite this, the indices of myeloma activity remained stable.Therefore, it remains unknown to us why the patient's disease remained stable for a 6-month period without myeloma-directed therapy, apart from zoledronic acid.
Multiple myeloma is characterized by osteolytic bone disease due to increased osteoclastic activity and reduced osteoblastic function, elements that constitute the predominant pathophysiological basis for the use of bisphosphonates in the disease process.The role of bisphosphonates in the management of multiple myeloma is to reduce the number of vertebral and non-vertebral fractures and osteolytic lesions. We hypothesize that the stabilization of our patient's disease during the 6-month period of cecal carcinoma treatment may be attributable to the continuation of zoledronic acid administration.
From the above it is inferred that patients should be informed about the risks of developing an SPM as a result of the therapy administered for the management of multiple myeloma.
Conclusions
Accumulating evidence supports an association between multiple myeloma and the development of a secondary primary malignancy (SPM), either attributable to treatment or as a result of the underlying disease process. Our patient was successfully managed surgically, received adjuvant treatment for cecal carcinoma, and discontinued antimyeloma therapy without complications being identified. The continued administration of bisphosphonates may have provided some protective effect against the myeloma.
Clinicians should be vigilant regarding the possibility of SPM development during the administration of immunomodulatory therapies or cytotoxic chemotherapies prior to autologous transplantation for the management of multiple myeloma.
- Secondary solid malignancies are a recognized late complication of myeloma treatment
- Prolonged immunomodulatory therapy increases colorectal cancer risk
- Iron deficiency anemia in myeloma patients should prompt colonoscopic evaluation
- Multidisciplinary coordination between surgical oncology and hematology is essential
- Colorectal cancer surveillance should be considered in long-term myeloma survivors
Sign in to join the discussion
Loading...